MDR for Pharmaceutical
The pharmaceutical sector operates across R&D labs, clinical trials, regulated manufacturing, and global supply chains. From molecule to market, every step relies on sensitive data, validated systems, and highly regulated processes. While this drives innovation and patient outcomes, it also creates new opportunities for cyber attackers.

Threats include state-sponsored espionage seeking intellectual property, ransomware halting batch release, and insider tampering with regulated records. Distributed sites, legacy OT, and rapid digitalisation all increase exposure. At the same time, regulators demand auditable proof of compliance with standards like ALCOA(+), ISO 27001, and FDA 21 CFR Part 11.Traditional monitoring is not enough.
Highly-skilled, Dedicated SOC
Continuous Monitoring
Expert Investigation
Incident Response
Pharmaceutical-Specific Risks We Address




The Smarttech247 Difference in Pharma MDR
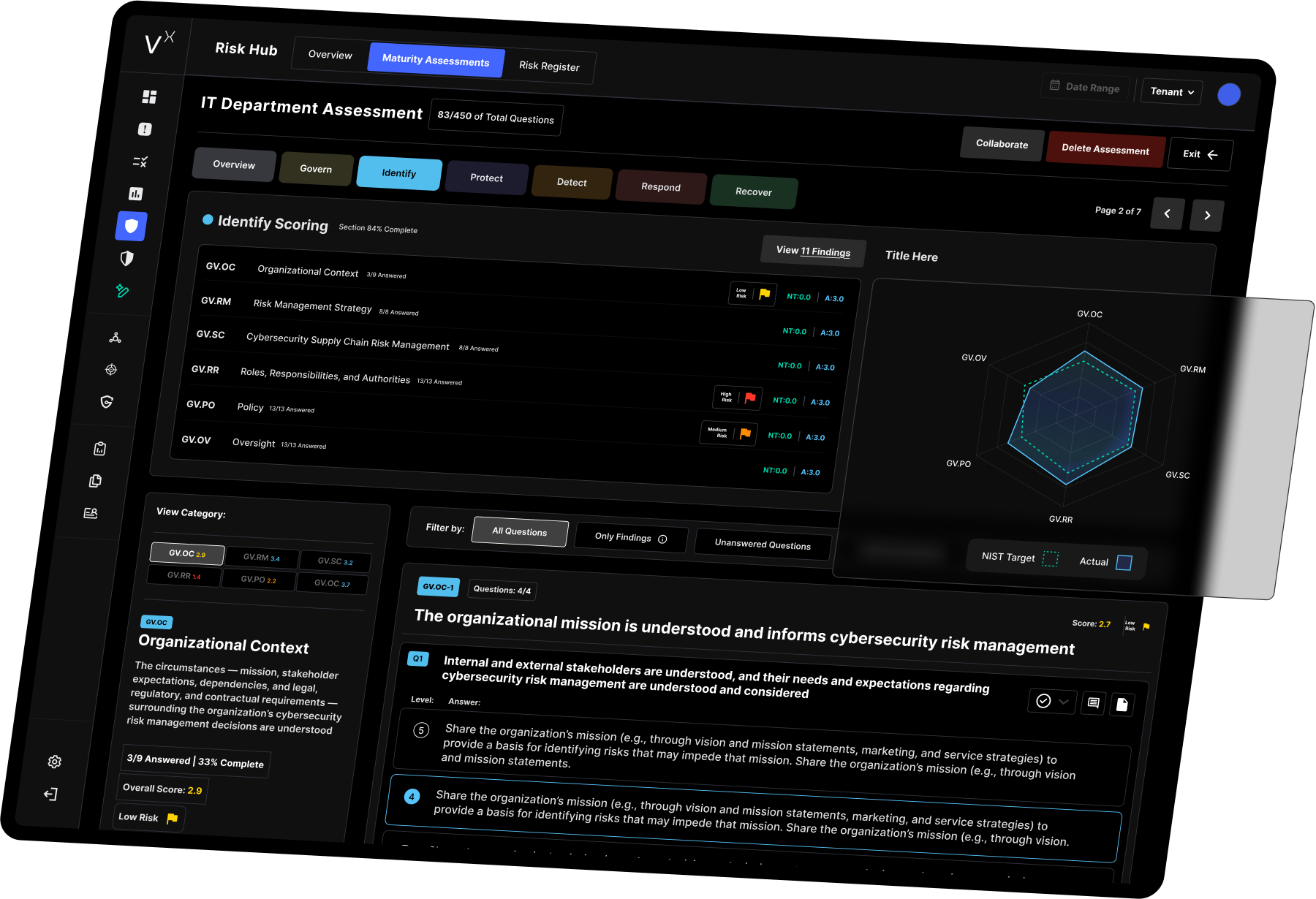
VisionX : The Platform Behind Smarttech247 MDR
Why VisionX Matters



Benefits of Smarttech247 MDR for Pharmaceutical
Our Expertise
We are a trusted MDR provider delivering high-quality, high-touch detection and response services worldwide. Our aviation-aware analysts bring a deep understanding of both operational and identity risks in the sector, ensuring tailored protection where it matters most.
Through integrated services such as Third-Party Risk Management, Digital Forensics, and Incident Response Planning, we provide end-to-end support that strengthens resilience. All of this is powered by our VisionX platform, which unifies data, reduces noise, and enables teams to focus on the incidents that truly matter.
Securing Pharma from Lab to Market
With Smarttech247 and VisionX, you gain round-the-clock protection, audit-ready evidence, and resilience across your entire pharma ecosystem.
Ready to scale your security and compliance operations?
We protect your on-premise/cloud/OT environments - 24x7x365






.jpeg)

